These results suggest that ATM acts as a promoter of tumorigenesis in cancer cells with overactivated Akt, and KU-55933 induces apoptosis and inhibits motility by blocking GLUT1-mediated glucose uptake and glycolysis in these cancer cells, which may lead to the use of KU-55933 and its analogs as new preventive or therapeutic agents against cancer. KU-55933 treatment also inhibits tumor growth and metastasis in vivo in mouse mammary tumors through inhibition of GLUT1 translocation and vimentin expression.

The roles of ATM in stimulating glucose uptake, glycolysis, motility, and proliferation of cancer cells were demonstrated by knocking-down ATM in these cells. We show that DOCK2, which is a mammalian homologue of Caenorhabditis elegans CED-5 and Drosophila melanogaster Myoblast City, regulates motility and polarity during neutrophil chemotaxis. Our results showed that while high concentration of glucose and insulin promote the expression of a mesenchymal biomarker (vimentin) in these cancer cells, KU-55933 strongly inhibits its expression as well as epithelial to mesenchymal transition. Neutrophil chemotaxis requires Rac activation, yet the Rac activators functioning downstream of chemoattractant receptors remain to be determined. Moreover, KU-55933 induces apoptosis and inhibits motility of cancer cells by inhibiting glucose uptake. KU-55933 also inhibits aerobic glycolysis and ATP production in these cells. In the present study, we found that aggressive breast and prostate cancer cell lines with overactivated Akt activity exhibit enhanced glucose uptake and GLUT1 translocation upon insulin treatment, and KU-55933, a specific inhibitor of ATM, inhibits insulin-mediated glucose uptake by blocking translocation of GLUT1 to the cell surface. However, the role of ATM in glucose uptake and tumorigenesis of cancer cells is unclear. We previously discovered that ATM, a protein kinase deficient in Ataxia-telangiectasia (A-T) disease, is an insulin-responsive protein that participates in insulin-mediated glucose uptake in muscle cells by stimulating glucose transporter 4 (GLUT4) translocation. Elucidation of the intrinsic signaling pathway of burbot spermatozoon spontaneous activation requires further study.Enhanced glucose uptake is coupled with elevated aerobic glycolysis (the Warburg effect) in cancer cells and is closely correlated with increased tumor aggressiveness and poor prognosis. Burbot spermatozoon activation occurs at 30 degrees C without modification of the spermatozoon membrane. No differences were found in protein profile and lipid composition with respect to activation of burbot spermatozoa at 30 degrees C. A role of swelling in activation of spermatozoa motility at 30 degrees C was rejected. No spermatozoon swelling was observed in non-ionic isotonic media at all studied temperatures. The result revealed that temperature increment from 4 to 30 degrees C increases burbot spermatozoa vulnerability to osmotic pressure, decreasing motility in extremely hypotonic media, and the presence of Ca2+ decreases swelling of spermatozoa. We explored spermatozoon swelling, lipid composition and proteome in relation to osmolality and temperature of swimming medium.
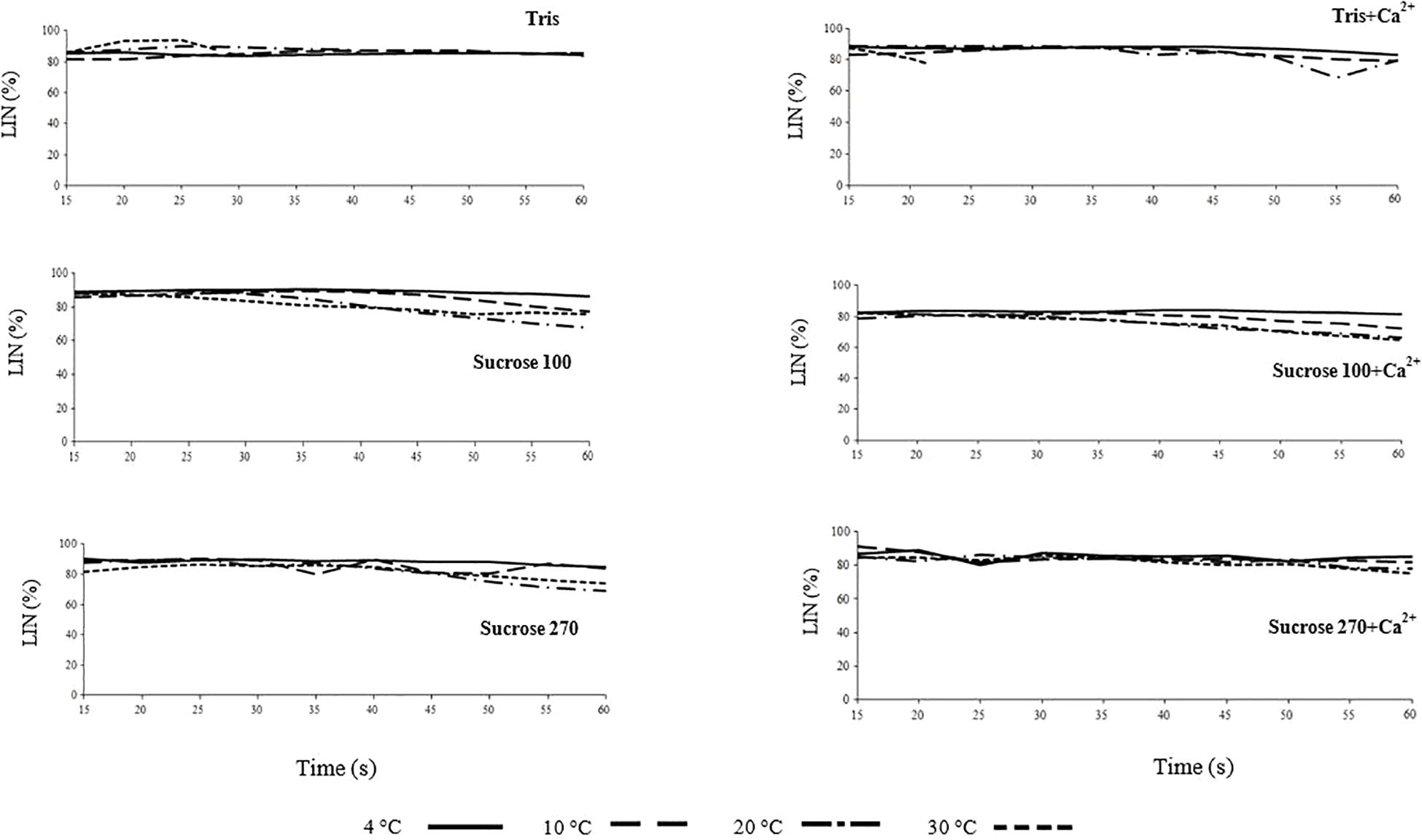
Despite available information on the roles of osmotic pressure, potassium (K+), and calcium (Ca2+) in activation of Eurasian burbot spermatozoon motility, the changes in milt biochemical composition and mechanisms underlying their activation at temperatures above optimal spawning temperature is still unclear.
